|
what's the diamond for food-grade ethanol look like pretty tame 
|
|
|
|
|

|
| # ? Jun 2, 2024 15:37 |
Zil posted:Reply from manufacturer: "Pour it down the drain." Erin Brockovich just felt a great disturbance in the force
|
|
|
|
|
Data Graham posted:Erin Brockovich just felt a great disturbance in the force Hell your local wastewater treatment plant is having a fit just imagining that poo poo hitting the water table.
|
|
|
|
Didn't they make a whole movie based on the town where everybody was dying from chromium VI in the groundwater?
|
|
|
|
|
M_Gargantua posted:Didn't they make a whole movie based on the town where everybody was dying from chromium VI in the groundwater? Yeah, "Erin Brockovich."
|
|
|
|
Notable for the epidemiology and toxicology not lining up to prove anyone had anything directly relatable to Cr-VI because its a lottery at the exposure levels in the community (in the background noise level of the exposure curve). On one hand this paints it as a NIMBY thing, on the other hand since it wasn't a closed loop when it could have been it was a homerun for them ignoring as low as reasonably achievable practices. Not to mention it got a lot of the actual malevolent bad actors with Cr-VI, like the dumping pickling/chroming liquor barely treated assholes, to take notice.
|
|
|
|
Over the past few days I suffered from repeated and prolonged exposure far in excess of recommended personal exposure limits.
|
|
|
|
Rozzbot posted:Over the past few days I suffered from repeated and prolonged exposure far in excess of recommended personal exposure limits. I have no particular knowledge about fluorocarbons, but apparently KFC is an effective antidote.
|
|
|
|
Speaking of Chromium (VI), I think I linked in here a while some guy who wanted to build a hybrid rocket in his backyard using ethanol fuel... and chromium trioxide as the solid-state oxidizer. E: yeah here it is Enourmo posted:hey guys i've never built a rocket before but for my first attempt I wanna use CrO3 for oxidizer May as well just post the OP: quote:[open sentence 1]Hello, most I know about rockets I learned from Scot Manly and "vintage space" on YouTube, most I know about chemistry I also learned from YouTube, and reading what not to do on MSDS papers, I have access to lots of chromium trioxide and ethanol, and I wanna build a Hipergolic reverse hybrid rocket with liquid fuel and solid oxidizer, this one I'm working on it tiny, the grain is 2 cm wide and like 10 cm long , and I have all the equipment and knowledge to do it safely (hazmat too) asside from it spewing cancer out the nozzle, is there anything I should consider whether this setup?[close sentence 1] I'll post photos when it's ready, but I just wanted to run the idea by this subreddit Fun fact, apparently the carcinogenic mechanism of Cr6+ is getting into your cell nucleus and forming heavy metal salts with your DNA. Fender Anarchist has a new favorite as of 10:43 on Mar 19, 2018 |
|
|
|
Seems about right for hexavalent chromium. Lead basically plops its fat rear end in any zinc channel it can find and doesn't leave. Something like that at least. Whatever screw biochem.
|
|
|
|
Mustached Demon posted:Whatever screw biochem. Hey, now - that's chromium VIs job!
|
|
|
|
Rozzbot posted:
Yup that's the stuff I used to make glazes with. Trick is to get it mixed *just* enough so the glassing agent coats the Chromium crystals before they start to decompose. (It tends to go clear if it's overheated because of side reactions with the glass) It's a tricky color to make since you only have a ~10deg C window to hit where it will go glassy and before the Chromium starts to break down.
|
|
|
|
Enourmo posted:Speaking of Chromium (VI), I think I linked in here a while some guy who wanted to build a hybrid rocket in his backyard using ethanol fuel... and chromium trioxide as the solid-state oxidizer. On the other hand, Cr(III) seems to have an important role in glucose metabolism, being part of a signalling complex formed when insulin binds to a cell wall. Cr(VI) specifically is bad, but chromium itself is an essential mineral.
|
|
|
|
All the talk of horrifying rocket propellents reminded me of this fun (fictional) story about "red mercury".
|
|
|
|
ATP_Power posted:All the talk of horrifying rocket propellents reminded me of this fun (fictional) story about "red mercury". That's a good read. Totally fictional of course, but good.
|
|
|
|
quote:rocket scientist who spent the 1950s in California and New Mexico, as a graduate student researching rocket fuels under John D. Clark 
|
|
|
|
The truth about Red Mercury is its own entertainingly insane story, though it sadly also leads to people trying to disassemble land mines.  The Curious Case of Red Mercury Campaign Against Red Mercury
|
|
|
|
Vavrek posted:The truth about Red Mercury is its own entertainingly insane story, though it sadly also leads to people trying to disassemble land mines. Here's another interesting not an explosive. https://en.wikipedia.org/wiki/Hafnium_controversy https://www.aps.org/publications/apsnews/200706/backpage.cfm Imaginary Weapons, Sharon Weinberger's book on this subject, is a fascinating read.
|
|
|
|
Old "educational" videos are a goldmine for terrible lab practices. https://www.youtube.com/watch?v=HGm80IFtbfA&t=196s https://www.youtube.com/watch?v=DhuuOWteelc&t=866s Queen_Combat has a new favorite as of 21:42 on Mar 25, 2018 |
|
|
|
Metal Geir Skogul posted:Old "educational" videos are a goldmine for terrible lab practices. Ah, a Jam Handy production! I recognized the narrator immediately. There are a whole lot of old educational, industrial and military films available if you have a Roku. Joe Screwdrivers Retro Tech Time Machine puts hundreds of them up for streaming. It saves having to search them out on YouTube.
|
|
|
|
This seems like it could potentially get pretty FOOF-y: https://www.nature.com/articles/d41586-018-03977-w Attempting to use AI to plan reactions for making new substances.
|
|
|
|
wolrah posted:This seems like it could potentially get pretty FOOF-y: https://www.nature.com/articles/d41586-018-03977-w That reminds me of a proposal I saw a while back; the idea was that with new developments in molecular sieves and chromatography and all that, we can isolate a molecule of anything from a mixture of reaction products, while the best method for creating new compounds is just "Throw some things in a beaker and wait for it to stop fizzing". So, why not take this to its full extent, and just throw everything in a vat to react together so we can isolate out interesting stuff later? The best part is that the whole vessel would proceed to equilibrium, so as we find and isolate anti-cancer drug #294, the vessel would produce more of it, while whatever muck we throw back in would be produced less. Will post it once I get home and can look it up.
|
|
|
|
If it's first drafts are pretty near an ideal synthesis candidate it's big news. But a lot of the more popular molecule churning softwares already had an index of possible pathway steps they could propose and it was a matter of getting the grad student to cross reference some of the roundabout or insane steps with something that made more sense. Robots have been churning chemistry bullshit since mainframes.
|
|
|
|
wolrah posted:This seems like it could potentially get pretty FOOF-y: https://www.nature.com/articles/d41586-018-03977-w Oh good, so Skynet can *tell* some chemist that they've stumbled across the ultimate artificial sweetener when in fact it's the world's deadliest delayed carcinogenic substance.
|
|
|
|
wolrah posted:This seems like it could potentially get pretty FOOF-y: https://www.nature.com/articles/d41586-018-03977-w Ignition posted:Just as Wharton was starting his IBA work, there occurred one of the weirdest episodes in the history of rocket chemistry A. W. Hawkins and R. W. Summers of Du Pont had an idea. This was to get a computer, and to feed into it all known bond energies, as well as a program for calculating specific impulse. The machine would then juggle structural formulae until it had come up with the structure of a monopropellant with a specific impulse of well over 300 seconds. Sorry for the hosed up formula, I'm phone posting, hopefully someone will correct it.
|
|
|
|
The Claptain posted:Sorry for the hosed up formula, I'm phone posting, hopefully someone will correct it.  quote this if you laugh every time
|
|
|
|
Enourmo posted:
H-C-NOpe 
|
|
|
|
Enourmo posted:
the definition of 
|
|
|
|
Enourmo posted:
So what kind of horrible side effects would this have on the human body? Or would it just be straight up blowing you up into neat piles of ash and body parts?
|
|
|
|
Enourmo posted:
So apart from the context and the fact it involves Fluorine, can someone talk me through exactly how horrible this is? Vincent Van Goatse has a new favorite as of 01:11 on Mar 31, 2018 |
|
|
|
That triple carbon bond looks so out of place.
|
|
|
|
Zil posted:So what kind of horrible side effects would this have on the human body? Or would it just be straight up blowing you up into neat piles of ash and body parts? I don't think that thing is subtle enough to have side effects. Everything it does is effects.
|
|
|
|
Vincent Van Goatse posted:So apart from the context and the fact it involves Fluorine, can someone talk me through exactly how horrible this is? I only did three semesters of chemistry in university, but my understanding is that the nitrogens want to be N≡N, and will become so as soon as someone looks at them funny, making all the oxygens, flourines and hydrocarbons blast out everywhere.
|
|
|
|
The NOF here stand for gently caress NO.
|
|
|
|
Enourmo posted:
|
|
|
|
Zil posted:So what kind of horrible side effects would this have on the human body? Or would it just be straight up blowing you up into neat piles of ash and body parts? I'm hoping that the dot above the right carbon atom is a chunk of cropped text and not a free electron somehow chilling on an alkyne group
|
|
|
|
Enourmo posted:
Is that a carbon radical? edit: can't be that would be a 9th electron
|
|
|
|
Arglebargle III posted:Is that a carbon radical? This is the free radical form. The stable form has a couple of covalent Fluorines ([F2]:2 hanging off that C using only van der Waals. That should stabilize this thing just fine.
|
|
|
|
Enourmo posted:
So does this thing explode if you A. look at it B. think about it C. the moment you write the formula down? e: by C, i mean does the diagram itself go boom. iospace has a new favorite as of 07:37 on Mar 31, 2018 |
|
|
|

|
| # ? Jun 2, 2024 15:37 |
|
Mustached Demon posted:The NOF here stand for gently caress NO. I don't remember much from the two chem classes I took in college (besides the hot girl sitting beside me wearing near-transparent Daisy Dukes), but yeah, even I know that much, and was going to make that joke if you hadn't.
|
|
|





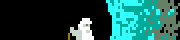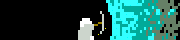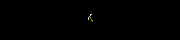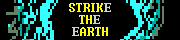

























 Bad Angus! Bad!
Bad Angus! Bad!