|
Luneshot posted:released heat of about 1000 watts per gram Holy exotherm, Batman! 
|
|
|
|

|
| # ? Jun 1, 2024 21:57 |
|
KozmoNaut posted:Holy exotherm, Batman! For when normal RTGs are too puny.
|
|
|
|
It reminds me of this entry on the TIWWW blog, which coincidentally is the one that named this thread:quote:http://pipeline.corante.com/archives/2010/02/23/things_i_wont_work_with_dioxygen_difluoride.php
|
|
|
|
KozmoNaut posted:It reminds me of this entry on the TIWWW blog, which coincidentally is the one that named this thread: I saw a mention of dioxygen chlorine trifluoride in that article's comments. That sounds like the most unholy molecule in existence that will kill you in any number of terrible ways just for looking at it wrong. We've already got a molecule that is evidence there is no such thing as a kind and loving god, let's cram some extra oxygens on the ends of it and see what happens 
|
|
|
|
Icon Of Sin posted:I saw a mention of dioxygen chlorine trifluoride in that article's comments. That sounds like the most unholy molecule in existence that will kill you in any number of terrible ways just for looking at it wrong. We've already got a molecule that is evidence there is no such thing as a kind and loving god, let's cram some extra oxygens on the ends of it and see what happens One of my favourites from TIWWW is the updates from Klap�tke, like the merely (hah) ridiculously explosive C2N14. Yup. Fourteen loving nitrogen atoms in a single molecule, with a couple of carbons acting like nightclub bouncers between two armoured divisions. It explodes if you so much as look at it funny. As in, "The sensitivity of C2N14 is beyond our capabilities of measurement. The smallest possible loadings in shock and friction tests led to explosive decomposition." It explodes if you try to get an infrared spectrum of it. Naturally, as eating a tube of Pringles on the next planet over will cause it to go "boom", its main use is in the field of getting rid of spectrometers you didn't like anyway.
|
|
|
|
His reverence for the sacred work of Klap�tke is endearing. They have to have their labs either underwater or in a hollow volcano, because they're clearly Bond villains.
|
|
|
|
KozmoNaut posted:It reminds me of this entry on the TIWWW blog, which coincidentally is the one that named this thread: That's gotta be per mole, not per individual reaction, because otherwise that's like nuclear energy.
|
|
|
|
Phanatic posted:That's gotta be per mole, not per individual reaction, because otherwise that's like nuclear energy. He clarifies later that that's per mol, yes.
|
|
|
|
ToxicFrog posted:He clarifies later that that's per mol, yes. Still nothing to sneeze at. I found a number for TNT of 616.4 kJ/mol, which translates to about 147 kCal/mol. So basically FOOF/H2S is about 3 times as potent as TNT.
|
|
|
|
Fucknag posted:Still nothing to sneeze at. I found a number for TNT of 616.4 kJ/mol, which translates to about 147 kCal/mol. Doesn't that blog also name a compound (prob also studied by Klap�tke), for which the description says that if you want to reduce its explosive qualities, it's best to dilute it with TNT? 
|
|
|
|
Carbon dioxide posted:Doesn't that blog also name a compound (prob also studied by Klap�tke), for which the description says that if you want to reduce its explosive qualities, it's best to dilute it with TNT? That's the nitrogen-cage abomination mentioned earlier. http://pipeline.corante.com/archives/2011/11/11/things_i_wont_work_with_hexanitrohexaazaisowurtzitane.php quote:Not that it's what you'd call a perfect compound in that regard - despite a lot of effort, it's still not quite ready to be hauled around in trucks. There's a recent report of a method to make a more stable form of it, by mixing it with TNT. Yes, this is an example of something that becomes less explosive as a one-to-one cocrystal with TNT. Although, as the authors point out, if you heat those crystals up the two components separate out, and you're left with crystals of pure CL-20 soaking in liquid TNT, a situation that will heighten your awareness of the fleeting nature of life.
|
|
|
|
That writing style
|
|
|
|
Has anybody ever done like, a general psych profile on chemists who specialize in explosives? I have to imagine there are some interesting common features there 
|
|
|
|
Being banned from Space Station 13 would probably be one of them.
|
|
|
|
no at least ss13 gives you the cushion of simulation, these are people who on occasion synthesize compounds capable of blowing off their limbs in the smallest amounts, by hand
|
|
|
|
'Hand' might be a bit generous.
|
|
|
|
i'll find it again later when i get off shift but i'm pretty sure tiwww has an example of a compound that's gotta be hand stirred for some hosed reason
|
|
|
|
Don't blame professional chemists, sometimes chemicals are invented by ten-year-old girls.
|
|
|
|
How the hell does stuff like that not get discovered sooner? Like shouldn't a room full of kids/a super computer "get" most chemicals?
|
|
|
|
PhazonLink posted:How the hell does stuff like that not get discovered sooner? Like shouldn't a room full of kids/a super computer "get" most chemicals? There are untold numbers of combinations of atoms and then we get into the insane math of their structure and pairing. Once you've put together something you can check and see if it's viable with some math, but marking off all the failures takes a lot of time. It's why protein folding crowdsourcing was popular a few years back, there's just so much stuff that any one thing could not possibly sift through it all with today's tech in any reasonable length of time.
|
|
|
|
Tollymain posted:i'll find it again later when i get off shift but i'm pretty sure tiwww has an example of a compound that's gotta be hand stirred for some hosed reason Dimethylcadmium, I think. The whole article is a good read, and his metaphors are exceptional as always. That's where "bored silly by something trying to kill you" showed up, at least. http://pipeline.corante.com/archives/2013/05/08/things_i_wont_work_with_dimethylcadmium.php quote:The general rule is, if you're looking for the worst organic derivatives of any metal, you should hop right on down to the methyl compounds. That's where the most choking vapors, the brightest flames, and the most panicked shouts and heartfelt curses are to be found. Methyl organometallics tend to be small, reactive, volatile, and ready to party. quote:If you have a nice wide spill of it, with a lot of surface area, you fool, it'll probably still ignite on its own, giving off plenty of poisonous cadmium oxide smoke. If for some reason it doesn't do that, you will still regret your decision: the compound will react with oxygen anyway and form a crust of dimethyl cadmium peroxide, a poorly characterized compound (go figure) which is a friction-sensitive explosive. I've no idea how you get out of that tight spot; any attempts are likely to suddenly distribute the rest of the dimethylcadmium as a fine mist. e: it was his article on metal azides. http://pipeline.corante.com/archives/2014/06/16/your_metal_azide_worries_are_over.php quote:And before making the metal azide in the first place, naturally, you need to ask "Do I want to blow myself to pieces?" That's because this isn't one of those set-it-and-forget-it Crock-Pot azide reactions. No, you're going to have to hand-craft these things: Icon Of Sin has a new favorite as of 17:40 on Jan 24, 2015 |
|
|
|
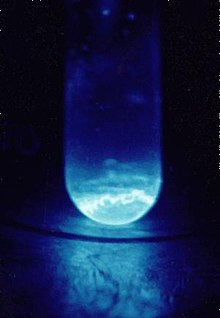
Luneshot posted:I particularly like the Wikipedia article for Einsteinium. No lie, I want to see this in person. How loving awesome.
|
|
|
|
7thBatallion posted:No lie, I want to see this in person. How loving awesome. Find a nuclear reactor to tour. They glow blue like that too.
|
|
|
|
DemeaninDemon posted:Find a nuclear reactor to tour. They glow blue like that too. Swimming in the pool at a nuclear reactor is on my bucket list. We've been through this a dozen times, and I'd no doubt be shot in the process. But it isn't Cherenkov radiation, is it?
|
|
|
|
7thBatallion posted:Swimming in the pool at a nuclear reactor is on my bucket list. We've been through this a dozen times, and I'd no doubt be shot in the process. Nope totally is Cherenkov.
|
|
|
|
DemeaninDemon posted:Nope totally is Cherenkov. Okay, now it really has my interest.
|
|
|
|
7thBatallion posted:Swimming in the pool at a nuclear reactor is on my bucket list. We've been through this a dozen times, and I'd no doubt be shot in the process. I got to photograph a nuclear power plant's refueling from the edge of the fuel basin, and it was ~~~magical~~~ You'll never see the blues I saw. Never
|
|
|
|
7thBatallion posted:Swimming in the pool at a nuclear reactor is on my bucket list. We've been through this a dozen times, and I'd no doubt be shot in the process. There is a way to get into the reactor pools to as a job. It involves scuba diving in the reactor vessel itself...where you've got all the normal hazards of diving (faulty air lines, decompression illness, etc), technical diving (underwater welding and working in general doesn't sound terribly appealing, though I hear the pay is decent) on top of the hazards of being near an active nuclear core. My buddy who works in a nuclear plant says these guys get their yearly allowable radiation dose within the first or second job of the year, depending on how long those jobs last and how often they're diving. http://www.popsci.com/science/article/2012-03/swimming-hot-side (Yes, this is a job I would consider applying to, at the nuclear plant not too far from here).
|
|
|
|
Platystemon posted:America can only answer this by doping bills with Americium. you probably have around a microgram or two of Americium in your house, so you can get right on this one today.
|
|
|
|
kastein posted:you probably have around a microgram or two of Americium in your house, so you can get right on this one today. Probably not recommended. 
|
|
|
|
Diving into a secured reactor pool is a great way of getting lead poisoning... ...well, insofar as kinetic introduction of lead to your body is concerned.
|
|
|
|
Minarchist posted:Diving into a secured reactor pool is a great way of getting lead poisoning... Already covered that. Hence why it's on my bucket list. As the last entry.
|
|
|
|
There's at least one, and I assume quite a few, pool-type reactors that are secured by all of a receptionist having to buzz you in. I've been through my university's research reactor building a few times and the main security checkpoint for it is the front desk where you leave your cell phone and pick up a visitor dosimeter badge. Their big security upgrade post-9/11 was to obscure it slightly by removing most of the blatant "Nuclear Reactor" signing and referring to it by a building number or acronym all the time.
|
|
|
|
Pitch posted:Their big security upgrade post-9/11 was to obscure it slightly by removing most of the blatant "Nuclear Reactor" signing and referring to it by a building number or acronym all the time. To be fair, that's probably a fairly effective security measure for the cost of its implementation. Can't hit it if no one knows it's there. 
|
|
|
|
Makes me want to open a bakery called NUCLEAR MATERIALS Lab and have a sale on yellow cake.
|
|
|
|
StandardVC10 posted:To be fair, that's probably a fairly effective security measure for the cost of its implementation. Can't hit it if no one knows it's there.
|
|
|
StandardVC10 posted:To be fair, that's probably a fairly effective security measure for the cost of its implementation. Can't hit it if no one knows it's there. The university research reactor I've been to uses the same techniques, but also has the building directory and evacuation maps fudged so the door that leads to it looks like it's for a basement janitor's closet.
|
|
|
|
|
Theris posted:The university research reactor I've been to uses the same techniques, but also has the building directory and evacuation maps fudged so the door that leads to it looks like it's for a basement janitor's closet. So, if an actual emergency happens, the firefighters get confused by the maps and everyone will die? Good security. At my university there was, to my knowledge, no research reactor (there used to be a particle accelerator, though). Funny thing was that the doors of the laboratory buildings with real expensive equipment were closed off. You either needed a key, or ring up a receptionist to open the door for you. Or simply use the elevated walkway from a next door building, the doors in those were always unlocked. Carbon dioxide has a new favorite as of 13:42 on Jan 25, 2015 |
|
|
|
7thBatallion posted:Makes me want to open a bakery called NUCLEAR MATERIALS Lab and have a sale on yellow cake. I've made yellow cake for my reactor physics dept on Groundhog Day, but the joke only makes sense if you think in English. :-/
|
|
|
|

|
| # ? Jun 1, 2024 21:57 |
|
Carbon dioxide posted:So, if an actual emergency happens, the firefighters get confused by the maps and everyone will die? Good security. I'm fairly sure the local fire department are made aware if there's a nuclear reactor in their jurisdiction.
|
|
|